|
Its position in modern periodic table will be(a) Group 1 and Period 3 (b) Group 2 and Period 3(c) Group 13 and Period 3 (d) Group 16 and Period 3 An element ‘X’ is forming an acidic oxide.Which of the following statement (s) about the Modern Periodic Table are incorrectThe elements in the Modern Periodic Table are arranged based on their decreasing atomic numberThe elements in the Modern Periodic Table are arranged based on their increasing atomic massesIsotopes are placed in adjoining group (s) in the Periodic TableThe elements in the Modern Periodic Table are arranged based on their increasing atomic number(a) (i) only(b) (i), (ii) and (iii)(c) (i), (ii) and (iv) (d) (iv) only.Give examples of alkaline metals present in Group-1 in the Periodic Table.(f) According to Newlands’ classification of elements, the properties of sulphur are similar to those of oxygen because sulphur is the _ element starting from oxygen. (b) The horizontal rows in a periodic table are called _. Fill in the following blanks with suitable words:(a) The basis for modern periodic table is _.How does the valency of elements vary in going down a group of the periodic table?.In which group of Mendeleev's periodic table is this element placed?(a) group II (b) group III (c) group V (d) group VIII Its most probable position in the modern periodic table is(A) Group 1 and Period 3 (B) Group 16 and Period 3 (C) Group 17 and Period 3 (D) Group 2 and Period 3 (a) What is the usual number of valence electrons and valency of group 18 elements of the periodic table?(b) What happens to the number of valence electrons in the atoms of elements as we go down in a group of the periodic table?.Consider the following elements:20Ca, 8O, 18Ar, 16S, 4Be, 2He Which of the above elements would you expect to be: (i) very stable? (ii) in group 2 of the periodic table? (iii) in group 16 of the periodic table?.(e) What are the advantages of the periodic table? (c) From the standpoint of atomic structure, what determines which element will be the first and which the last in a period of the periodic table? (d) Explain why, the properties of elements are repeated after 2, 8, 18, and 32 elements in the periodic table. (a) What is a group in the periodic table? In which part of a group would you separately expect the elements to have (i) the greatest metallic character (ii) the largest atomic size?(b) In what respects do the properties of group 1 elements differ from those of group 17 elements? Explain with examples by taking one element from each group.Write an example of a compound formed with(a) chlorine (Group 17 of Periodic Table)(b) oxygen (Group 16 of Periodic Table) Carbon, Group (14) element in the Periodic Table, is known to form compounds with many elements.(a) How does the electropositive character of elements change on going down in a group of the periodic table?(b) State how the valency of elements varies (i) in a group, and (ii) in a period, of the periodic table.The periodic table of elements is a table of chemical elements ordered based on their atomic number, electron configuration, and recurring chemical properties. Saturated Unsaturated and Supersaturated.Reaction Quotient and Le Chatelier's Principle.Prediction of Element Properties Based on Periodic Trends. 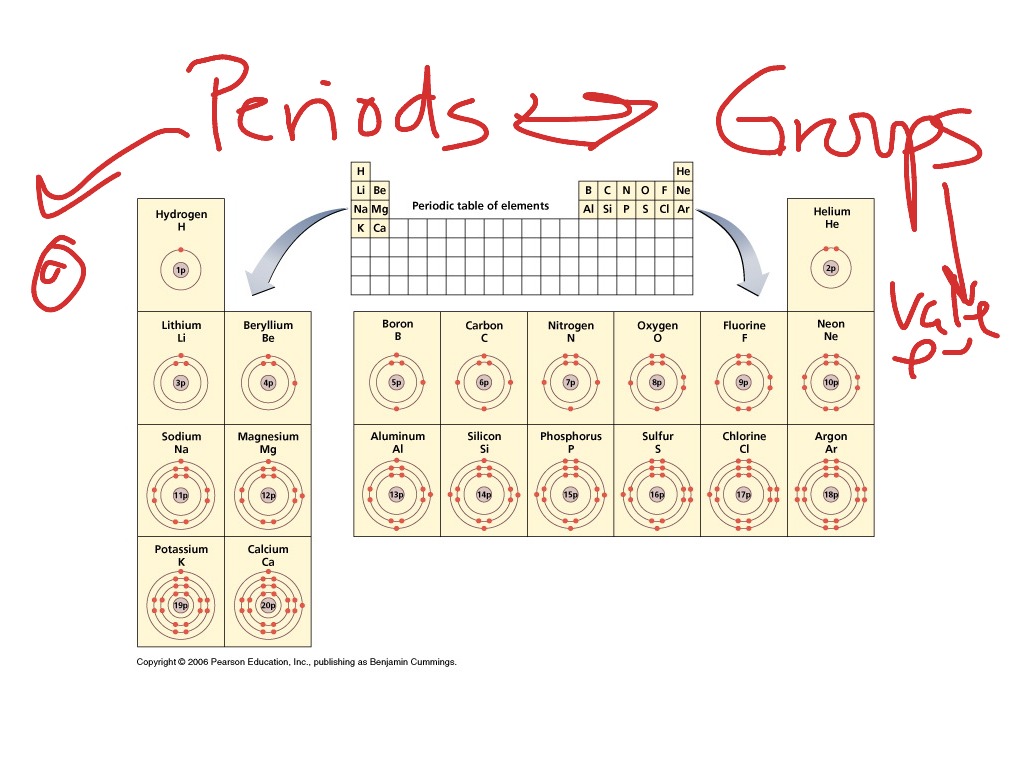
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
